The Definitive Bioavailability Guide for Pharmaceutical and Nutraceutical Formulators
If you are formulating calcium supplements, fortified foods, or pharmaceutical preparations for the European or global market, you have likely defaulted to calcium carbonate for its cost efficiency and high elemental calcium content. But the science of calcium absorption tells a more nuanced story, and choosing the wrong calcium source can mean your end consumers absorb significantly less calcium than your label promises. In this guide, we break down the clinical evidence, formulation trade-offs, and sourcing considerations that will help you make the right calcium selection for your next product, backed by over 30 years of citrate manufacturing expertise at our WHO-GMP certified facility.
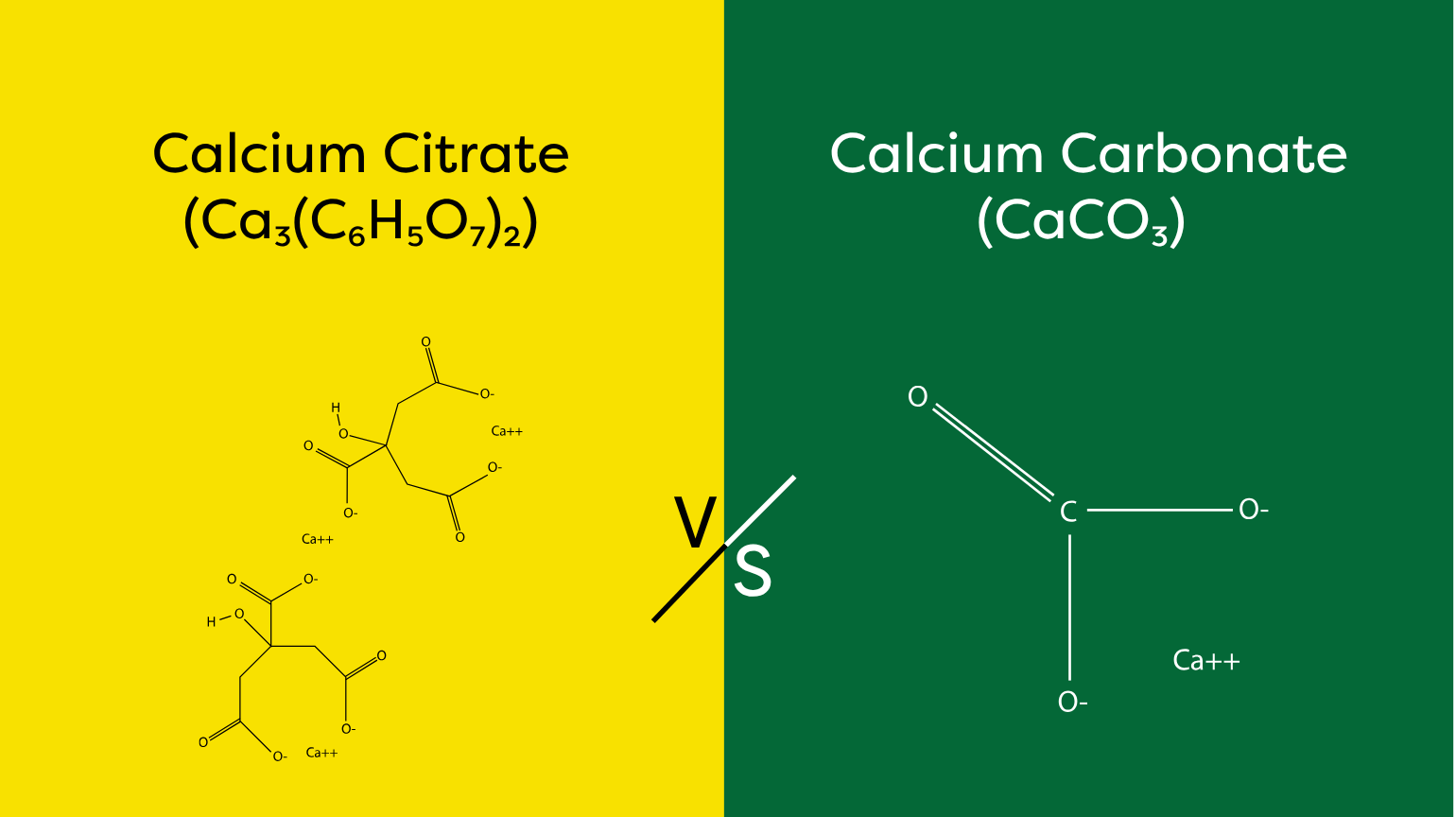
What Is the Difference Between Calcium Citrate and Calcium Carbonate?
Calcium citrate and calcium carbonate are the two most widely used forms of supplemental calcium, differing in their chemical structure, elemental calcium content, absorption mechanism, and clinical suitability. Understanding these differences is essential for pharmaceutical companies, nutraceutical brands, and food manufacturers who need to select the right calcium source for their formulations.
Calcium carbonate (CaCO₃) is a salt derived from limestone, marble, or oyster shells. It contains approximately 40% elemental calcium by weight, making it the most calcium-dense supplement form available. A 1,250 mg tablet delivers 500 mg of elemental calcium. However, calcium carbonate is an insoluble salt that requires hydrochloric acid in the stomach to dissolve and release its calcium ions for absorption.
Calcium citrate (Ca₃(C₆H₅O₇)₂) is the calcium salt of citric acid. It contains approximately 21% elemental calcium by weight, meaning a higher mass is needed to deliver the same elemental dose. However, calcium citrate is a soluble salt that dissolves readily in water and does not depend on stomach acid for absorption. This property makes it the preferred form for populations with reduced gastric acid production, and it is why demand for pharmaceutical-grade calcium citrate is accelerating among European supplement manufacturers.
At the same time, the distinction extends beyond chemistry. Calcium carbonate is best absorbed when taken with food, as the meal stimulates gastric acid secretion. Calcium citrate can be taken with or without food, offering greater dosing flexibility. At Daffodil Pharmachem, we manufacture both forms, but we have observed a significant increase in calcium citrate demand from our European pharmaceutical and nutraceutical clients over the past three years, driven precisely by this clinical flexibility.
Why Does Calcium Bioavailability Matter for Pharmaceutical Formulations?
Calcium bioavailability is the percentage of ingested calcium that is actually absorbed into the bloodstream and made available for physiological use, and it directly determines the clinical effectiveness of any calcium supplement or fortified product. A supplement that delivers 500 mg of elemental calcium on the label but only achieves 20% absorption provides just 100 mg of usable calcium to the patient.
For pharmaceutical companies formulating calcium products for the European market, bioavailability is not merely a marketing claim. The European Food Safety Authority (EFSA) recommends a Population Reference Intake (PRI) of 950 mg/day of calcium for adults aged 25 and above. Achieving this target through supplementation requires selecting a calcium form that maximizes the ratio of absorbed calcium to ingested calcium.
Moreover, bioavailability becomes even more critical in specific patient populations. In the EU alone, approximately 4.3 million new fragility fractures occurred in 2019, driven in large part by osteoporosis and inadequate calcium absorption.
In addition, the growing use of proton pump inhibitors (PPIs) across Europe further compounds the problem. PPIs suppress stomach acid production, which directly impairs calcium carbonate absorption but does not affect calcium citrate. For formulators targeting aging populations or patients on chronic PPI therapy, calcium citrate offers a bioavailability advantage that calcium carbonate cannot match. This is one of the key reasons our pharma-industry clients increasingly specify calcium citrate in their formulation briefs.
What Does the Clinical Evidence Say About Calcium Citrate vs Calcium Carbonate Absorption?
A landmark meta-analysis published in the American Journal of Therapeutics concluded that calcium citrate is absorbed 22% to 27% better than calcium carbonate, whether taken on an empty stomach or with meals. This study, conducted by Sakhaee et al. at the University of Texas Southwestern Medical Center, analyzed 15 clinical studies involving 184 subjects.
Absorption on an Empty Stomach
Five studies involving 71 subjects measured calcium absorption when supplements were taken without food. In this fasting condition, calcium citrate demonstrated 27% higher bioavailability than calcium carbonate. This difference is clinically significant because calcium carbonate requires stomach acid for dissolution, and acid production is minimal in a fasting state.
Absorption With Meals
Six studies involving 65 subjects measured absorption when calcium was taken alongside food. Even with meal-stimulated acid secretion, calcium citrate still showed 22% superior absorption. This finding disproves the common assumption that taking calcium carbonate with food fully compensates for its acid-dependent absorption pathway.
Recent Clinical Confirmation (2024)
A double-blind, randomized crossover trial published in Surgery for Obesity and Related Diseases in 2024 further confirmed calcium citrate’s superiority. The study involved 150 post-bariatric surgery patients and found that calcium citrate significantly lowered PTH levels and demonstrated enhanced relative bioavailability.
Specifically, calcium citrate achieved a higher AUC of 76.1 mg/dL per hour versus 74.7 mg/dL per hour for carbonate, and urinary calcium excretion was significantly greater at 83.7 mg versus 68.6 mg over 9 hours.
Earlier Foundational Research
An early comparative study in the Journal of Clinical Pharmacology found that calcium citrate was 2.5 times more bioavailable than calcium carbonate when given with a meal. A follow-up study reaffirmed these findings and added evidence that calcium citrate’s PTH-suppressing effect may contribute to bone loss prevention in postmenopausal women.
As a manufacturer that has been producing high-purity calcium citrate for over three decades, we at Daffodil Pharmachem have seen this clinical evidence translate directly into procurement decisions. European pharmaceutical companies that once sourced calcium carbonate exclusively are now adding calcium citrate to their formulation portfolios specifically because of these proven absorption advantages.
How Do Calcium Citrate and Calcium Carbonate Compare Across Key Formulation Parameters?
Pharmaceutical and nutraceutical formulators must evaluate calcium sources across multiple dimensions beyond bioavailability. The table below provides a direct comparison based on published pharmacopeial and clinical data.
| Parameter | Calcium Citrate | Calcium Carbonate |
| Chemical Formula | Ca₃(C₆H₅O₇)₂ | CaCO₃ |
| Elemental Calcium | 21% by weight | 40% by weight |
| Bioavailability | 22–27% higher than carbonate | Baseline reference |
| Stomach Acid Required | Not required | Required for dissolution |
| Absorption Timing | With or without food | Best with food only |
| GI Side Effects | Lower incidence | Higher (gas, bloating, constipation) |
| PPI User Suitability | Recommended | Reduced absorption |
| Older Adults (65+) | Preferred | Less suitable |
| Post-Bariatric Surgery | Clinically preferred | Not recommended as primary |
| Pharmacopeial Grades | USP, EP, BP, FCC | USP, EP, BP, FCC |
| EU Additive Code | E333 | E170 |
| REACH Registration | Required >1 tonne/year | Required >1 tonne/year |
| Cost Per g Elemental Ca | Higher | Lower |
Daffodil Pharmachem’s calcium citrate is REACH registered, removing a significant compliance barrier for European procurement teams.
 When Should Formulators Choose Calcium Citrate Over Calcium Carbonate?
When Should Formulators Choose Calcium Citrate Over Calcium Carbonate?
Calcium citrate is the superior choice when the target consumer has compromised stomach acid production, requires dosing flexibility, or needs maximum calcium absorption per serving.
Supplements for Aging Populations (65+)
Adults over 65 commonly experience reduced gastric acid secretion. Research indicates that up to 30% of adults over age 60 have significantly reduced stomach acid levels. For this population, calcium carbonate absorption drops substantially, while calcium citrate absorption remains unaffected.
In Europe specifically, approximately 22 million women and 5.5 million men had osteoporosis in 2019, and this number is projected to increase. Formulators who can offer clinically validated, high-bioavailability calcium citrate products have a clear competitive edge in this demographic.
Formulations for Patients on Proton Pump Inhibitors
PPIs such as omeprazole and pantoprazole suppress hydrochloric acid production, directly impairing calcium carbonate dissolution. The AAFP has noted that calcium citrate may be better absorbed than other calcium salts for patients taking acid-reducing medications.
Renal Therapeutics and Urinary Alkalinisation
Calcium citrate serves a dual therapeutic function in renal health. Beyond calcium supplementation, citrate acts as a urinary alkaliniser, inhibiting calcium oxalate stone formation. At Daffodil Pharmachem, we produce both potassium citrate and calcium citrate in pharmaceutical grades, allowing formulators to source complementary renal ingredients from a single WHO-GMP certified supplier.
Food and Beverage Fortification
Calcium citrate’s water solubility makes it suitable for fortifying beverages, dairy alternatives, and liquid nutritional products. Our food-grade calcium citrate is produced under FSSC 22000 and HACCP standards, making it directly suitable for European food and beverage applications.
When Does Calcium Carbonate Remain the Better Choice?
Calcium carbonate is the preferred choice when cost efficiency, tablet size minimisation, and high elemental calcium density are the primary formulation priorities, and the target consumer has normal gastric acid levels.
Cost-Sensitive Mass Market Supplements
For basic supplementation targeting healthy adults under 50, calcium carbonate delivers more elemental calcium per tablet at a lower cost. Calcium carbonate accounts for approximately 46.9% of the global calcium supplement market.
Antacid Combination Products
Calcium carbonate’s ability to neutralise stomach acid makes it uniquely suitable for combination antacid-supplement products. This application has no equivalent in calcium citrate formulations.
Solid Food Fortification
In bakery products, cereals, and confectionery, calcium carbonate integrates well into dry formulations without affecting taste, texture, or shelf stability.
That said, many of our European clients take a portfolio approach: they use calcium carbonate for cost-sensitive mass-market products and source calcium citrate from Daffodil Pharmachem for their premium, evidence-based, and condition-specific formulations. This dual-source strategy serves both market tiers without compromising clinical efficacy where it matters most.
How Daffodil Pharmachem Supports European Calcium Citrate Sourcing?
Sourcing pharmaceutical-grade calcium citrate for EU distribution requires verification of manufacturing certification, pharmacopeial compliance, REACH registration, and supply chain traceability documentation that meets EudraLex Volume 4 requirements. Daffodil Pharmachem meets and exceeds each of these requirements.
Our Certifications and EU Compliance
As Asia’s largest WHO-GMP certified citrate manufacturer, Daffodil Pharmachem holds every certification that European procurement teams require:
- WHO-GMP certification for our pharmaceutical manufacturing facility (the only plant of its scale in Asia)
- REACH registration under EU Regulation (EC) No 1907/2006, enabling direct import into the European market
- FSSC 22000 for food safety management systems
- ISO 9001:2015 for quality management systems
- ISO 14001:2015 for environmental management systems
- BRC Global Standard for food safety
- HACCP certification for hazard analysis and critical control points
- Halal and Kosher certifications for multi-market product flexibility
- USP, EP, BP, and FCC grade compliance verified by Certificate of Analysis for every batch, with purity levels of 99%+
Our Sustainability Edge and CBAM Readiness
With the EU’s Carbon Border Adjustment Mechanism (CBAM) expected to expand to chemical imports by 2027 or 2028, Daffodil Pharmachem’s sustainability practices give our EU clients a measurable compliance advantage:
- 50% solar-powered manufacturing at our 500,000 sq. ft. facility
- 100% water recycling across all production processes
- ISO 14001:2015 certified environmental management system
- Working toward net-zero emissions, with verified carbon footprint data available for CBAM reporting
Production Capacity and Reliability
Our facility produces over 80 metric tonnes of high-purity citrate ingredients daily using advanced SCADA-controlled reactors and automated systems. For European buyers, typical lead times to major EU ports (Hamburg, Rotterdam, Antwerp) are 4 to 6 weeks. Priority scheduling with reduced production lead times of 5 to 7 business days is available for time-sensitive orders.
Direct-from-Manufacturer Pricing Advantage
By sourcing directly from Daffodil Pharmachem, European pharmaceutical companies typically achieve 10% to 20% lower per-unit costs compared to distributor channels. Beyond pricing, direct sourcing offers shorter documentation chains, faster CoA turnaround, and direct access to our quality assurance and regulatory affairs teams.
To discuss calcium citrate specifications, request a sample, or receive a Certificate of Analysis, contact our team directly.
Key Takeaways
- Calcium citrate delivers 22% to 27% higher bioavailability than calcium carbonate, whether taken on an empty stomach or with meals, according to a meta-analysis of 15 clinical studies.
- Calcium carbonate contains 40% elemental calcium by weight, while calcium citrate contains 21%. Calcium carbonate requires fewer tablets per dose but absorbs less efficiently.
- Calcium citrate does not require stomach acid for absorption, making it the preferred form for older adults, PPI users, and post-bariatric surgery patients.
- Osteoporosis affects approximately 22 million women and 5.5 million men in the EU, driving demand for calcium ingredients with proven bioavailability.
- The global calcium supplement market is valued at USD 4.6 billion in 2024 and projected to reach USD 9.4 billion by 2034.
- EU-ready sourcing requires WHO-GMP, REACH registration, and pharmacopeial compliance (USP/EP/FCC), along with verified sustainability data as CBAM expands to chemical imports.
- Daffodil Pharmachem produces over 80 metric tonnes of pharmaceutical-grade calcium citrate daily at Asia’s largest WHO-GMP certified citrate facility, with REACH registration, FSSC 22000, Halal, Kosher, ISO 9001, ISO 14001, and BRC certifications, supplying brands in 25+ countries.
What Steps Should European Formulators Take Next?
European pharmaceutical and nutraceutical companies should audit their current calcium formulations against the bioavailability evidence, evaluate their target demographics, and align their sourcing strategy with evolving EU regulatory requirements.
- Review your product portfolio’s calcium source against target demographics. If your supplements target adults over 50, PPI users, or post-surgical patients, evaluate switching to calcium citrate.
- Request Certificates of Analysis from your current calcium supplier. Verify purity levels meet pharmacopeial standards (99%+ for USP/EP grade).
- Verify REACH registration status. Confirm your supplier holds a valid REACH registration. Daffodil Pharmachem’s REACH registration covers our pharmaceutical and food-grade calcium citrate products.
- Evaluate your supplier’s sustainability credentials. Begin collecting verified carbon footprint data and energy source breakdowns now.
- Consider direct-from-manufacturer sourcing. Compare total cost of ownership against distributor channels.
- Explore combination formulations. Daffodil Pharmachem produces both magnesium citrate and sodium citrate in pharmaceutical grades, allowing formulators to source a complete citrate portfolio from a single certified supplier.
Conclusion
Calcium citrate and calcium carbonate both play important roles in the global calcium supplementation landscape, but they are not interchangeable. The clinical evidence is clear: calcium citrate delivers 22% to 27% superior bioavailability, does not require stomach acid for absorption, causes fewer gastrointestinal side effects, and is the clinically preferred form for aging adults, PPI users, and post-surgical patients.
For European formulators, the choice should be driven by clinical evidence and target population needs, not raw material cost alone. As regulatory requirements around REACH, CBAM, and sourcing transparency tighten, partnering with a WHO-GMP certified, REACH-registered calcium citrate manufacturer with verified quality and sustainability credentials is no longer optional. It is a competitive necessity.
Daffodil Pharmachem is ready to be that partner. With over 30 years of citrate manufacturing expertise, 80+ metric tonnes of daily production capacity, and every certification the European market demands, we supply the calcium citrate that helps leading global brands deliver on their label promises. Request a sample or CoA today.